Introduction
Sinonasal metastasis from distant extranasal sites is extremely rare. Nearly half of metastatic sinonasal malignancies originated from the kidney, followed by lungs, breasts, urogenital ridge, gastrointestinal tract, thyroid and pancreas.1) Hepatocellular carcinoma (HCC) has a marked predilection for metastasis due to the characteristic route of angioinvasion.2) Usually, it metastasizes to lungs, lymph nodes and musculoskeletal system.3) On the other hand, metastasis in the paranasal sinuses is not common, especially only 12 cases of metastasis into sphenoid sinus have been reported in the English literature. The incidence of metastasis only to the sphenoid sinus is even more unlikely. We report a case of successful management with radiotherapy for isolated sphenoid sinus metastatic HCC presenting intractable nasal bleeding, which failed with several surgical hemostasis.
Case Report
A 62-year-old man was referred to the department of otorhinolaryngology for recurrent epistaxis during a month. Anterior nasal packing performed in the emergency room was failed several times. He also had progressive right visual loss for one week prior to visit.
The patient has been suffered from liver cirrhosis with genotype 1b hepatitis C and was heavy alcohol abuser who consumed 20 standard drinks a day. During follow-up, he was diagnosed with HCC three months ago. In computed tomography (CT) with contrast, well enhanced 8 multiple hepatic masses (maximum size: 5.8 cm) with expanding nodular pattern were observed. However, vascular invasion, lymph node enlargement and distant metastasis were not found. Moreover, there was no evidence of portal hypertension on CT scan. The patient underwent transcatheter arterial chemoembolization once.
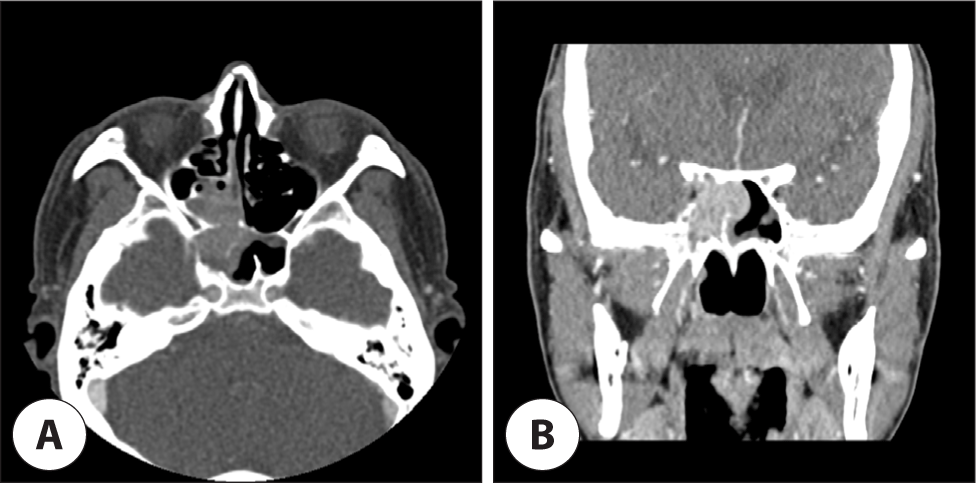
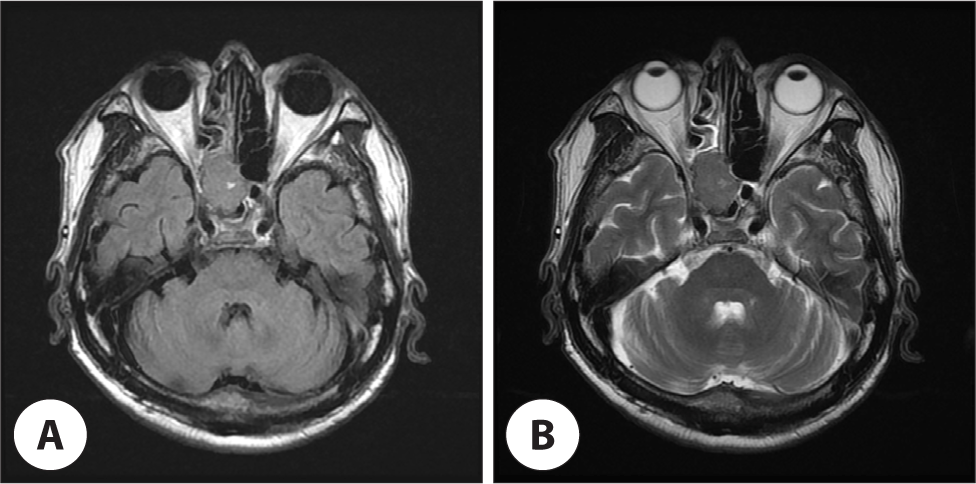
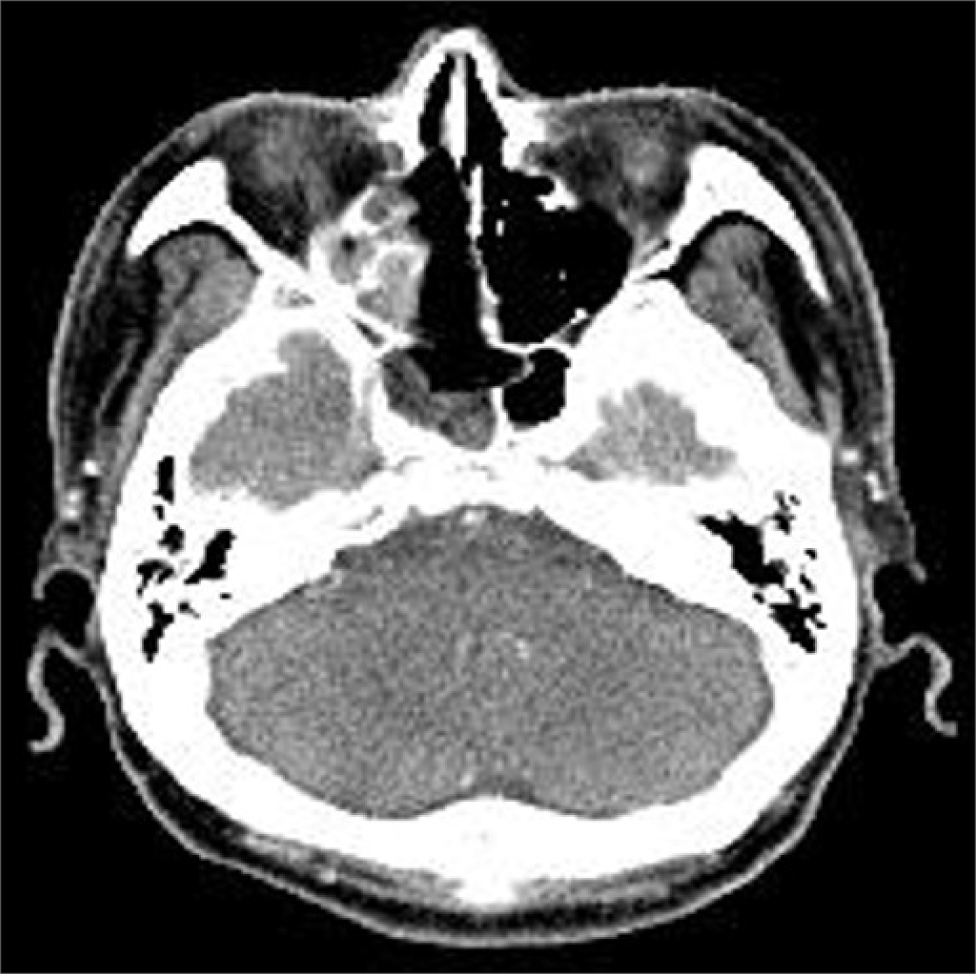
Nasal bleeding was observed at the right sphenoethmoidal recess in the nasal endoscopic examination, which was thought to be derived from polypoid mass (Fig. 1). The coagulation profile test was normal, except for a slightly decreased platelet count of 111,000/µL. Paranasal sinus CT demonstrated homogeneous enhancing solid mass filling the right posterior ethmoid and sphenoid sinus cavity. And leakage of some contrast media was observed, suggesting intralesional bleeding. There was a bony erosion of right optic canal medial wall and right sphenoid sinus roof by mass with extension of optic apex (Fig. 2). The ill-defined mass identified in magnetic resonance imaging (MRI) appeared to press the right optic nerve and was also seen to be separated from the posterior wall of sphenoid sinus and the brain parenchyma (Fig. 3).
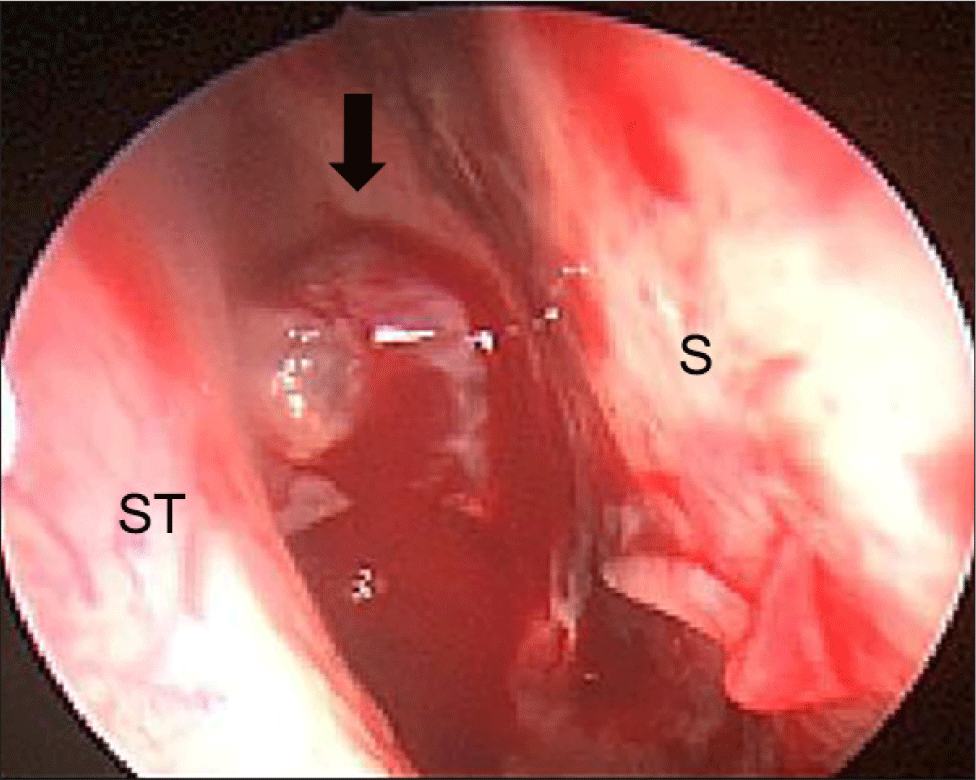
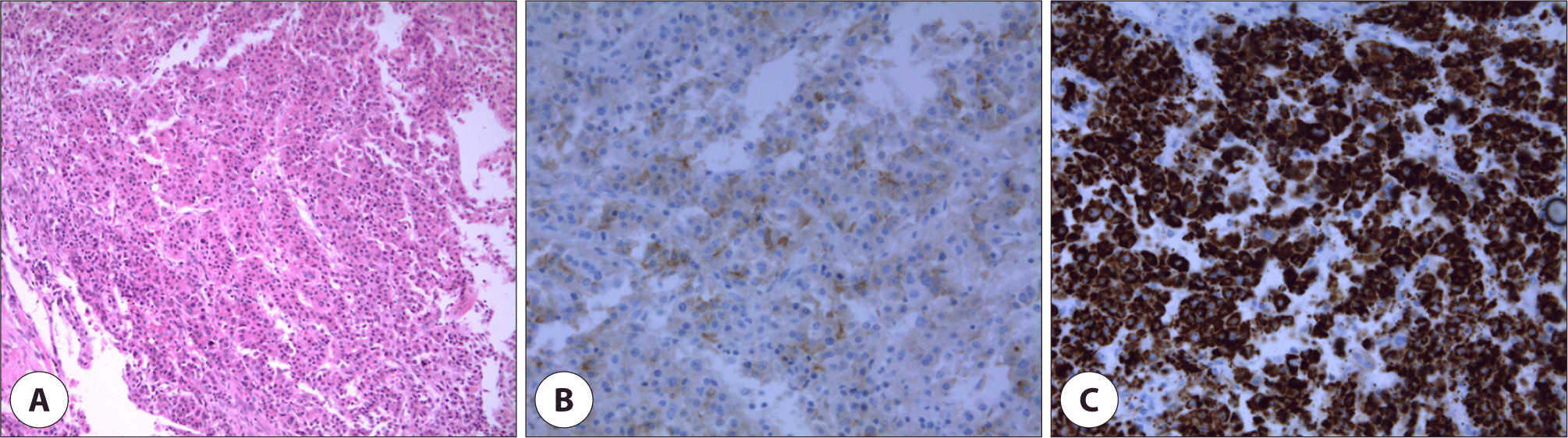
The patient underwent endoscopic sinus surgery for decompression of right optic nerve and biopsy. Intraoperatively, the mass seemed to be originated from the superolateral wall of right sphenoid sinus and grew out toward sphenoethmoidal recess. Histopathologic examination revealed the characteristic of HCC with malignant cell proliferation of canalicular pattern in hematoxylin and eosin stain. And immunohistochemistry showed positive for glypican-3 and hepatocyte specific antigen (Fig. 4). Intranasal packing was performed using Surgicel®, Gelfoam® and Merocel®, but intractable epistaxis was occurred after removal of packing materials in the second post-operative day. Angiography was performed for vascular embolization because epistaxis was uncontrolled with additional anterior nasal packing. Active bleeding was observed from the branch of the meningohypophyseal trunk of right internal carotid artery in angiography. So, the embolization was canceled due to concerns about complications such as infarction. Emergency operation were performed to bleeding control, active bleeding were stopped after changing Merocel® to Nasopore Forte®. Postoperative facial b. simulation CT showed remnant tumor in right sphenoid sinus (Fig. 5). Palliative radiotherapy (40 Gy in 16 fractions for 4 weeks) was planned as a treatment option after consultation to radiologic oncologist. Fortunately, epistaxis was successfully controlled after thirteen fractionated radiotherapy. Subsequently, the patient refused additional treatment for primary and metastatic malignancy, and was follow-up loss after discharge.
Discussion
The metastasis of HCC into the sphenoid sinus is very rare, thought to be due to the specificity of the metastatic pathway. The exact mechanism by which HCC metastasizes to paranasal sinus area is not known yet. In general, the metastatic route of HCC can be classified as hematogenous or lymphogenous spread.4) The most apparent route is via the caval venous system, then through the pulmonary circulation, thence from the heart to the sinuses through the arterial vessels of the head and neck.5) But, since this pathway refers to the possibility of metastasis through the pulmonary circulation, it seems insufficient to explain metastasis into the sinuses without lung metastasis. Instead, the vertebral venous plexus was assumed as an alternative route for the spread of cancer cells.6) There is the existence of free communication between the neck, thorax, abdomen and pelvis venous systems with the non-valve vertebral venous plexus that extends from cranial base to coccyx. Any pressure increment inside the abdomen can create an ascendant flow through the vertebral venous plexus, and cancer cells may thus spread to paranasal sinuses. Finally, using the lymphogenous route, tumor emboli from the regional lymph nodes may flow into the thoracic duct and can reach the head and neck.
The symptoms for metastatic sinonasal tumors are non- specific and related to the characteristics of the primary tumor and the metastatic sites.1) HCC metastasis appears as a hypervascular mass, mainly represented by epistaxis.2) Although epistaxis may be related to a decrease in the liver function depending on the origin site of disease, but hypervascularity is considered to be a more reasonable cause. In addition, invasion into the sphenoid sinus is associated with symptoms such as headache, proptosis, diplopia, or vision changes.7) In our case, epistaxis was accompanied by diplopia and impaired vision, but laboratory test results for coagulation except platelet count were normal.
The prognosis of metastatic HCC in the head and neck is generally poor. Unfortunately, HCC is relatively resistant to chemotherapy resulting unsatisfactory outcomes. Palliative management for advanced HCC could be selected to reduce neurologic sequelae and relieve symptoms when the meaningful therapeutic options are not available.8) Surgery may provide some opportunity for patients to increase the survival rate by reducing the tumor burden and to also achieve a better quality of life by alleviating symptoms such as epistaxis or nasal obstruction. However, classically, surgery is not routinely considered for metastatic disease. Especially, the infiltrative nature of the metastatic HCC at sphenoid sinus can be difficult to obtain complete surgical resection with clean margins. One consideration for palliative control of epistaxis is embolization of feeding vessels by angiograpy.9) Most sinonasal tumors receive blood supply from external carotid artery branches, which would be amenable to embolization. But, in our case, embolization was not performed due to concerns about complications such as infarction since feeding artery was a branch of internal carotid artery.
Recent study showed that radiotherapy may contribute to the control of intracranial tumor and the prevention of intracranial hemorrhage in patients with brain metastasis from HCC.10) In addition, a refractory bleeding at solitary mandibular metastasis of HCC successfully treated by palliative radiotherapy was reported.11) Radiotherapy has been also used to palliate bleeding from other malignant tumors such as lung, cervical, gastric, and bladder cancers.12) Radiation causes denudation of the intima of blood vessels that supplies the tumor, leading to capillary necrosis and thrombosis, which blocks the lumen of blood vessels, achieving hemostasis.13) And also the role of radiation therapy in hemostasis is thought to be a reduction in tumor size and induction of fibrosis in the peripheral osseous structure.11)






